Hydrus® Microstent: Minimally Invasive Glaucoma Treatment
The Hydrus® Microstent is a minimally invasive FDA-approved implantable device for the treatment of open angle glaucoma. The Hydrus® is a Micro-Invasive Glaucoma Surgery (MIGS) approved to be done in conjunction with cataract surgery. Since it is done at the same time as cataract surgery, no additional incisions are necessary.
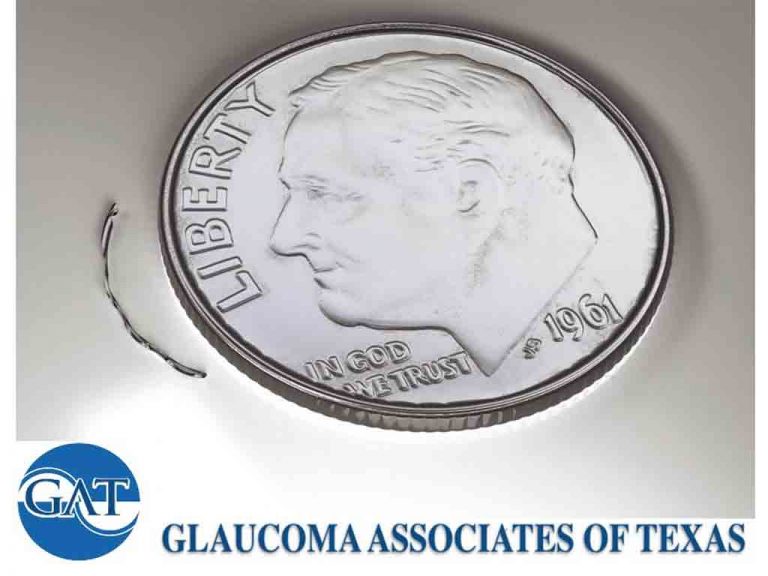
The surgical procedure involves placement of an 8mm nitinol (a nickel and titanium alloy) implant through the trabecular meshwork and into Schlemm’s canal to decrease intraocular pressure via 3 mechanisms: 1) bypassing the trabecular meshwork, 2) dilating Schlemm’s Canal, and 3) spanning over 3 clock-hours (more than any other implantable MIGS device). These mechanisms combine to enhance aqueous outflow from the anterior chamber and lower intraocular pressure.
Although the Hydrus® is made of nitinol (an alloy metal), patients can safely undergo an MRI scan in most circumstances; please contact our office with any questions or visit the Hydrus® Information page.
This procedure does not involve opening-up the conjunctival tissues to access Schlemm’s canal. This is a major advantage of this procedure as it makes future glaucoma surgery (if needed) a lot simpler with less risk of failure.
Who is a Good Candidate for the Hydrus® Microstent?
Doctors use Hydrus® for the surgical treatment of mild to moderate open-angle glaucoma. They avoid using it in patients with primary or secondary angle-closure glaucoma, neovascular glaucoma, traumatic glaucoma, uveitic glaucoma, or anterior chamber angle anomalies.
How Does the Hydrus® Microstent Work?
The Hydrus® Microstent reduces IOP by combining three mechanisms:
- Increasing aqueous outflow by bypassing the trabecular meshwork allowing aqueous to go directly into Schlemm’s Canal.
- Dilating Schlemm’s Canal to enhance aqueous outflow
- Spanning 3 clock-hours of Schlemm’s Canal, thereby reliably reducing intraocular pressure.
For more details visit the Hydrus® website.
What Steps Does a Surgeon Take During a Hydrus® Microstent Procedure?
When you and your doctor decide to proceed with a Hydrus® Implant, our preoperative scheduler will provide detailed instructions on preparing for surgery and reaching the operating room. See Preoperative instructions for more information.
Doctors perform this outpatient procedure in an ambulatory surgery center. The surgery usually takes less than 30 minutes, though you will stay at the center for several hours. Surgeons typically use local anesthesia with intravenous sedation. After surgery, they cover the eye with an eye patch and protect it with a plastic shield overnight. The next morning, your ophthalmologist removes the patch and shield and examines the eye. Then, they prescribe eye drops to prevent infection and reduce inflammation. For more details, click on Postoperative instructions.
For several weeks following the surgery, your ophthalmologist will observe your eye closely and examine you frequently. It will take several weeks after your surgery for the healing to be complete. During this time, it is not unusual for your intraocular pressure, as well as vision, to fluctuate. You will be ready to change your glasses prescription at around 4-6 weeks after surgery.

 DONATE NOW
DONATE NOW